Scientific developments at Attoworld have led to spawning a new research institution. In 2019, the Hungarian Ministry of Innovation and Technology approved financial support for a national project to propel health monitoring based on new laser technology. For this purpose, the Center for Molecular Fingerprinting (CMF) was founded in Budapest. Although still a very young institution, the CMF is about to acquire a status of being one of the National Laboratories in Hungary and is relevant to the research efforts at the LMU.
In the springtime of its evolution, what are the main aims of CMF?
It has been known that a few molecules in our blood can be regarded as reporters which are beneficial to track if we suffer from specific diseases. But is there more to that?
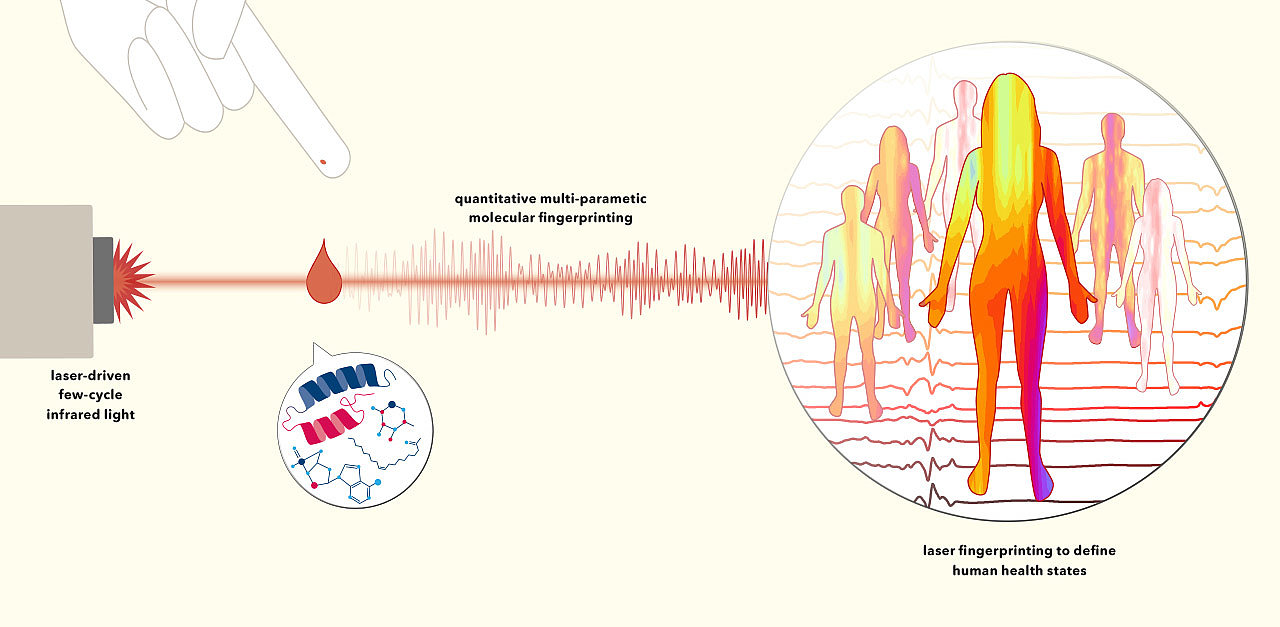
Can one imagine the day when a doctor will tell us with confidence whether we are still fully healthy, simply by analysing as little as a drop of our blood? In other words: Could looking at signals from all the different circulating molecules together – most of which are unknown to date or still impossible to quantify – in a combined pattern be accounted for comprehensive monitoring of human “healthiness” without the need of physically examining each and every organ by different, sometimes invasive or even noxious interventions? Unfortunately, major health problems in their preliminary stage run under the radar of current detection techniques too often, as the improvement of early disease markers is tedious and slow.
Suffice to say that we have started to pave the path towards thislong anticipated goal! Scientists of the CMF-LAP collaboration are challenged to devise a pioneer blood-based diagnostic assay for health monitoring in a fast and accurate manner, further advancing recently developed mid-infrared laser technology (Pupeza et al., Nature 577, 52, 2020). Our laser scientist are working on electric-field resolved laser spectrometry that is capable of sensing the minuscule changes in the full molecular composition of blood, which herald a disease already before it manifests physical symptoms.
How could one possibly detect minor deviations from health by analysing a drop of blood?
The idea we pursue is to use the newly available technology to attribute the health status of a person to the information embedded in the molecular composition of a blood sample. Blood not only nurtures our cells and tissues, but it also interconnects each segment of the human body and carries soluble molecules released by organs, which are often indicative of their function. Thus, when a local anomaly emerges within our body, say in the liver for example, a severe liver disease could possibly be prevented if having a routine method at hand that quickly analyses the overall molecular content of blood during a regular health-check. In this case the key to detecting e.g. liver disease is that the analysis needs to be sensitive and broadband enough to cover a wide range of molecular substances (or classes of molecules) – to reflect the early pathophysiological changes of the liver. There are immense efforts exerted by basic biomedical researchers and the pharmaceutical industry to tap on the footprints of diseases in the blood. Although these endeavours have resulted in an ever-increasing number of diagnostic tests being used in daily practise, the number of tests, which actually make it from the research bench to the market, is surprisingly small, and the pace of developing new tests is erratic.
A late diagnosis of a serious health condition often leads to a much worse outcome. In fact, we are still often faced with way too late disease detection! So, it is pivotal to switch paradigm and opt for a sensitive pan-molecular detection of diseases, and even pre-disease states, to better guard our health. It has become clear that such interventions must be performed in a personalized manner since every individual’s blood constitution is different at the molecular level. Equally, even every person’s healthy state is a little bit different. Practically speaking, following-up a person’s current health status by our proposed assay regularly might become paramount for timely-detecting relevant deviations.
There are ways to sensitively capture blood composition by detecting a diversity of molecules at once
Besides variations in the constitution, it is understood that the quantity of certain molecules in the blood vary within a healthy reference range. We often get the heat going through our veins when we look at our blood test results and notice that a particular value is out of the indicated reference range, or near the thresholds. Often this is ahead of our notion of being actually ill. It has been increasingly accepted that one single molecular biomarker can hardly be used for accurate prognosis of any complex disease (apart from a few very specific cases of microbial infections or specific metabolic disorders of the kidneys or the liver). Instead, molecular profiling has been growing recent attention in important fields, like the most common diseases as cardiovascular diseases and cancers. While looking at a bigger picture, such blood analysis can be interpreted as putting a perplexing puzzle together – where we do not even have a clear idea of how the final picture looks like or sometimes, how a single piece should appear. Ideally, we would have a complete “quantitative blood-based molecular landscape” which identifies all biomarkers and estimates their concentration. In reality, we are speaking of several tens of thousands of circulating molecules that have currently been associated to have an instructive role in medical diagnostics. Yet, there is no such technology to screen all types of molecules at once, or which is sensitive enough to detect all the important ones in the complex mixture of blood.
 Human blood samples to be processed for infrared spectroscopy. Picture: Thorsten Naeser
Human blood samples to be processed for infrared spectroscopy. Picture: Thorsten Naeser
The scientists at LAP have recently demonstrated the first generation of such a technology that allows for higher sensitivity in reporting on the nature of the entire complexity of molecules in liquid water-based samples (Pupeza et al., Nature 577, 52, 2020). Our paradigm relies on new ultrashort laser-pulsed field-resolved infrared spectroscopy measuring the molecular fingerprint that the sample leaves in the infrared, characteristic of both the nature and the quantity of the constituting molecules. We went on and devised new ways how to derive such infrared molecular fingerprints of human blood, after the blood and immune cells swimming therein are removed. In this sense, field-resolved infrared molecular fingerprints capture signals dependent on chemical bonds within all circulating molecules, independently of the higher-order structure of molecules in the sample – thus probing complex human samples in a pan-molecular manner that has not been imaginable with other technologies. Human biofluids will now be efficiently characterized by this new approach, with molecular fingerprinting on a new level of sensitivity combined with molecular coverage.
 High-thoruput blood serum/plasma handling using automated liquid sampling system. Picture: Thorsten Naeser
High-thoruput blood serum/plasma handling using automated liquid sampling system. Picture: Thorsten Naeser
Is that sufficient, and could we now go on and apply infrared molecular fingerprinting to capture diseases or characterize and monitor different states of human health?
The technology, while being extremely promising, is still in its early state and has been awaiting the clinical applicability. Nevertheless, it is prime time to get the feet wet and test our assay on real-world settings, which is the monitoring of healthy individuals. In collaboration with different clinics at the LMU in Munich, LAP scientists have already shown that every person has a unique infrared fingerprint. Importantly, our scientists could already provide evidence that these blood-based infrared fingerprints are stable over time and robust to daily perturbations. The existing data suggest that the infrared molecular fingerprints are, in principle, applicable to monitoring the deviations from a healthy state. Only if robust signal patterns can be derived out of further large-scale independent studies, can we make sure that one day we will be able to distinguish between healthy and disease electric-field molecular fingerprints which will be our entry pass to clinical practice.
How will CMF efforts contribute to health monitoring?
CMF realizes that turning the vision into reality requires continued dedicated technological development along with well-controlled populational studies at the forefront of the scientific research. It starts from the development of new mid-infrared laser sources via advancing sample handling and detection technologies and goes towards finding optimal data acquisition and processing strategies. Over the last couple of months, a new team has formed at CMF with the particular goal to tackle these technical and scientific challenges. In close collaboration, the CMF laser development and technology team at the Garching research center is joining forces with LMU laser scientists to combine all technological advances in a one of a kind prototype device for molecular fingerprinting of blood.
How is CMF planning to collect, store and analyze populational studies?
CMF has currently been setting up large-scale populational studies to register blood-based infrared molecular fingerprints from individuals over time. A network of collaborations across Hungary is being built, serving the aim of collecting several tens of thousands of blood samples and corresponding clinical information from every participant, with a first visit to collect a baseline and with several follow-up visits. This entails following the participants when their regular medical check-up is due for many years to come. Why the follow-up visits? As noted above, although molecules of blood trail differ from person to person, infrared molecular fingerprints of a particular person stay rather stable over time with the premise that no disease is being developed. To be in a position to figure out most precisely whether a healthy person is indeed healthy, it is paramount to compare a person’s most recent fingerprint with his/her own earlier fingerprints. CMF will create a framework to recruit volunteers that will donate blood samples twice yearly, followed-up over several years.
 Viola Zóka at the LEX laboratory preparing blood-based samples for cryogenic storage. Picture: Thorsten Naeser
Viola Zóka at the LEX laboratory preparing blood-based samples for cryogenic storage. Picture: Thorsten Naeser
A further advantage of blood-based human sample analysis is that samples can be stored deep-frozen. What is more, if stored under cryogenic conditions at –185ºC, they can be securely preserved for decades. For this goal to achieve, a new fully automatic liquid nitrogen-based biorepository will be built at CMF, which will store hundreds of thousands of little tubes filled with blood samples (with no cells present). This resource will fuel the development of molecular fingerprinting andallow to evaluate its clinical utility in years and decades to come.
Additionally, CMF is planning to build new laboratories in Budapest – to be in the position to collect, prepare, store, measure and analyze samples at the same time, under the same roof. Once the laser laboratories of CMF will open their gates, the field-resolved infrared molecular fingerprinting will be fundamental to devise and evaluate high-throughput approaches for monitoring human health. Moreover, the corresponding infrared molecular fingerprints will be analyzed together with medical data using several emerging artificial intelligence techniques.
What is the perspective in seeing deeper without the need of looking deeper?
Although almost two centuries have passed since Dr. Elisabeth Blackwell, the first US female doctor, spotlighted the value of monitoring human health “we are not tinkers who merely patch and mend what is broken… we must be watchmen, guardians of the life and health of our generation, so that stronger and more able generations may come after …”. Ever since the unmet medical needs remained the same, but the diagnostic approach is now driven by laser technology that can detect individual chemical bonds – thus letting us see through to the bottom of the molecular zoo in our blood! The transfer from basic scientific understanding about blood-based infrared molecular fingerprints to the applied question of what these fingerprints will tell us about diseases and conditions will probably take several more years to come. In the meantime, the CMF team, with its headquarters office at Budapest and scientific teams at Garching and Budapest, is further strengthening and extending the research efforts, that have already taken a deep dive into the depths of field-resolved molecular fingerprinting.
 The CMF team in Budapest is working hand in hand with their colleagues in Munich (Nov. 4th 2020, Budapest). Picture: Károly Dobrosi
The CMF team in Budapest is working hand in hand with their colleagues in Munich (Nov. 4th 2020, Budapest). Picture: Károly Dobrosi